
Civilization rests on concrete more than any other material. And we’ve been making it for thousands of years. But even so, a new study suggests that “greener” concrete is possible that may save almost half of its tremendous cost in energy and carbon dioxide.
Concrete is an artificial stone, made by binding gravel and sand, known as aggregate, together with the compound known as cement. Cement is like bread dough in that it consists of a small set of ingredients in highly variable proportions. In the case of cement, they are silicon, calcium, and water. Cement is not a set of specific calcium-silicate minerals, but a half-crystalline, half-glassy mush or gel called C-S-H (for “calcium silicate hydrate”). There appears to be no one ideal formula for C-S-H.
Cement is expensive in terms of energy and greenhouse gases. It must be manufactured at very high temperatures by roasting various minerals. And beyond that, the calcium comes from limestone (calcium carbonate), which releases carbon dioxide from its own substance in the roasting process and adds to the greenhouse-gas burden. Given that cement accounts for as much as 10 percent of global carbon emissions, it’s worth learning how to minimize this cost and make concrete as green a product as we know how.
The ancient Greeks and Romans made concrete structures that survive to this day. After all these centuries, we have lots of experience making cement. But apparently trial and error, even systematic experimentation, hasn’t been enough.
An open-access paper published last month in Nature Communications used a novel method to systematically explore the C-S-H recipe. A team at MIT’s Concrete Sustainability Hub, led by Roland Pellenq, used a computer to build atom-by-atom models of cement from a wide range of recipes, then predict the mechanical strength of the resulting material. It was a kind of brute-force approach varying only the ratio of calcium to silicon, or C/S, between 1 and 2, that is, a formula with equal parts calcium and silicon and a formula with half as much calcium. The genius of their method was that the computer could build many different molecular models of cement from the exact same formula, then calculate the strengths of the various models.
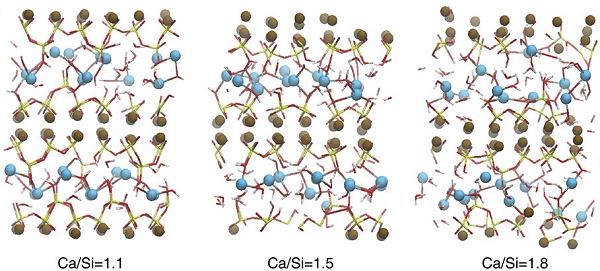
The next step was to search for the strongest models, which Pellenq’s team found at a sweet spot near a C/S ratio of 1.5. Certain models with that composition were one-third to one-half as much stronger. The key to the added strength appears to be a more subtle feature of the models, the medium-range correlation length of the silicon and calcium groups. If we can learn to control these features, we can emit less carbon by using less cement overall.
