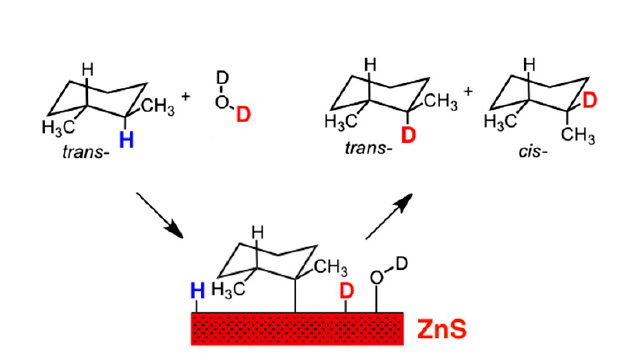
A research team in Arizona has found that common zinc ore may have a key role as a catalyst—a chemical magic wand—in the complex chain that must have led to the start of life on Earth. Their new paper in the Proceedings of the National Academy of Science (PNAS) shows that the widespread mineral sphalerite acts as an unexpected catalyst with organic matter, adding a new power tool to the workshop that gave rise to life.
How did the mineral world bootstrap itself into the first forms of life? This is the question propelling a burgeoning area of research.
Ordinary chemistry is little help. Creationists always argue that you can’t turn a bunch of dead chemicals into living things by random chance. That’s correct as far as it goes. The key is to start with a high-energy hydrothermal setting, where everything is more chemically active. Most of the basic ingredients of life—organic compounds based on carbon—are formed and transformed in very hot places. A few kilometers below the Earth’s surface, as well as at “black smoker” vents on the seafloor, temperatures are well above the boiling point and the necessary chemical reactions of organic compounds are possible. Oil and natural gas, for instance, are organic remains of once-living things that have been transformed in the hot depths.
Lab experiments at hydrothermal conditions generally reproduce the right chemical reactions, but the reactions happen way too slow. The trick that life has mastered is to rely on enzymes—chemical shortcuts using catalysts. Enzymes are proteins that include catalytic metal atoms in strategic places. Something in the exact configuration of the metal in the enzyme molecule fits a target molecule as a key fits a lock, and a reaction can happen rapidly that otherwise would take forever.
Enzymes are well and good, but they’re complicated molecules that raise the same objection as life itself. Life had to arise with the help of catalysts that are simpler than locks and keys. So origins-of-life researchers have been seeing what various ordinary minerals can do in hot water full of organic material. This is where the new PNAS paper fits in.