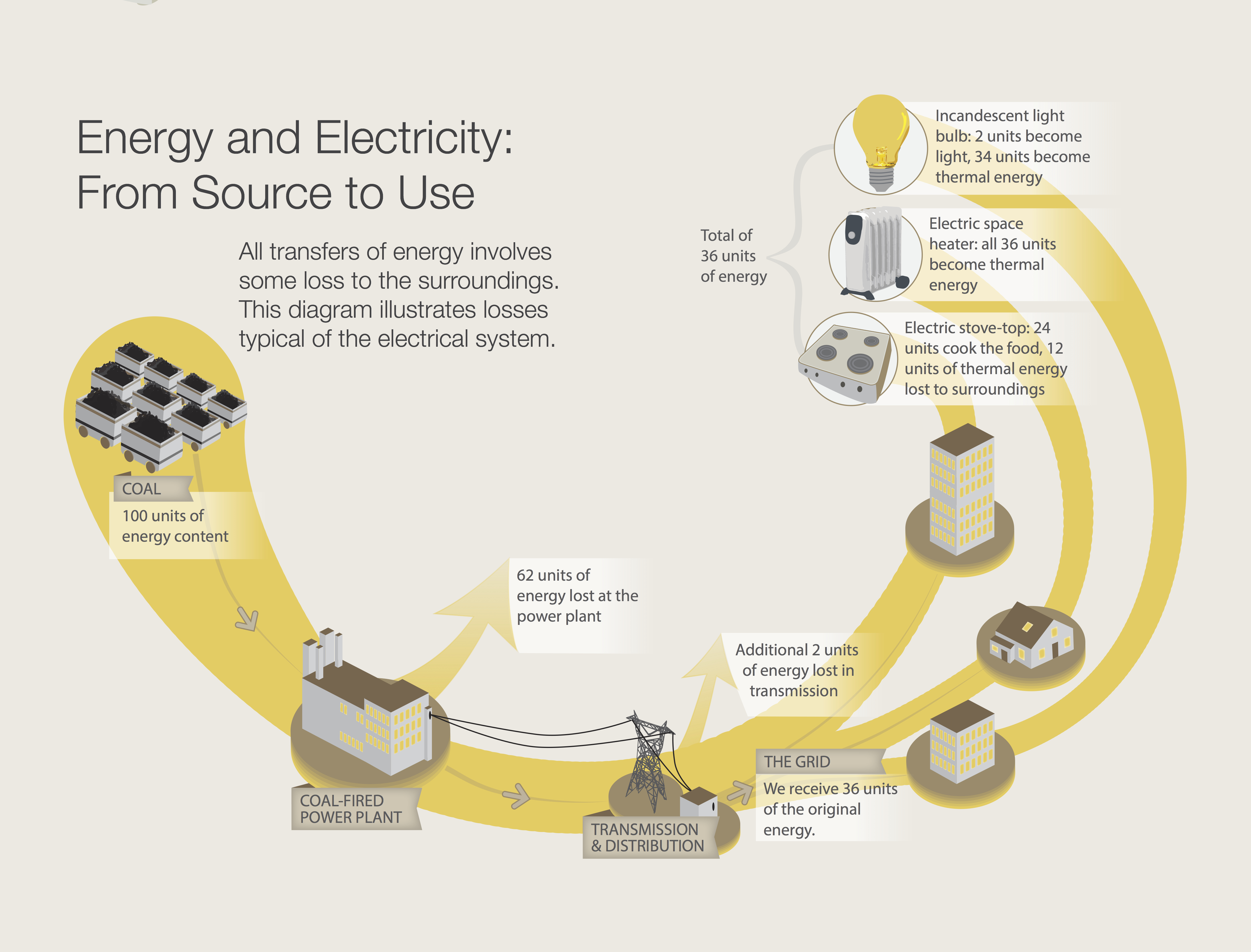
Energy can be transferred from one object to another. Energy can also be transformed from one form to another. As these actions happen, some of the energy is “lost” to the surroundings. This lost energy becomes thermal energy. This fact is known as the Second Law of Thermodynamics, which says that it is not possible to transfer or transform energy with 100 percent efficiency.The diagram above illustrates losses typical of the electrical system.
Another law of physics, the law of conservation of energy, says that energy can’t be created or destroyed. It can only be transferred and transformed. This can be confusing when you think about how often you’re asked to conserve energy — turn off lights, drive less, insulate buildings and so on. If there is a law of physics that says energy will be conserved no matter what we do, then why worry? The amount of energy in the universe isn’t going to change.
It turns out that although the total amount of energy in the universe remains constant, the total amount available for human activities isn’t constant. Humans transfer and transform energy from resources in the environment in very intentional ways for very specific purposes.
These energy resources can be nonrenewable resources, such as fossil fuels and nuclear energy, or renewable resources, such as solar, wind, water, geothermal, and biomassenergy. Nonrenewable energy resources are available in limited supply. Using less of, or conserving, these energy resources means they will be available for use for a longer period of time.
This infographic is featured in our Energy e-book series. Click on the thumbnails below to download our free e-books or subscribe to our iTunes U course. You can also visit our e-books page to view our other offerings.